Pre-qualification seen as significant step towards self-sufficiency in healthcare in continent where more than 70% of drugs are imported
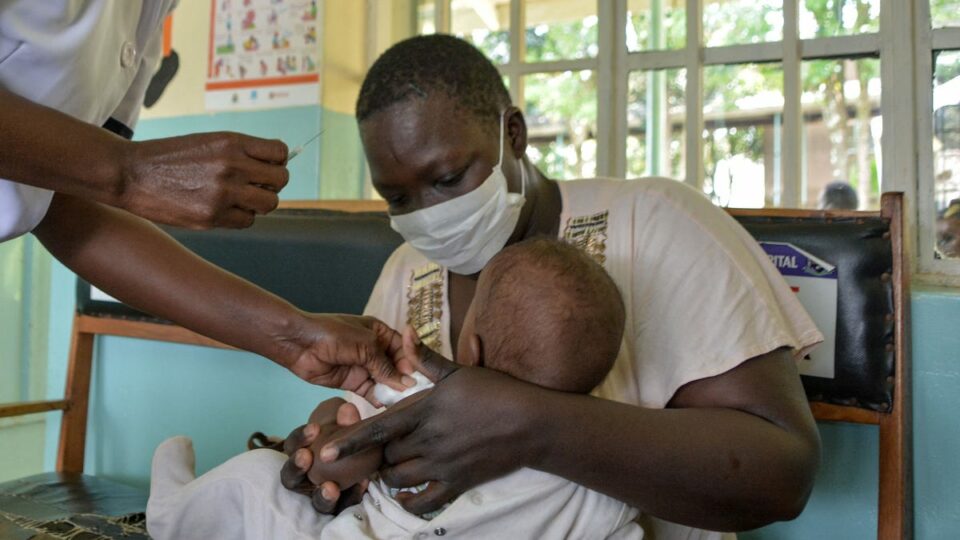
A Kenyan pharmaceutical company, Universal Corporation Limited, has become the first manufacturer in Africa to receive World Health Organization (WHO) approval to produce a lifesaving malaria drug.
The antimalarial drug, sulfadoxine-pyrimethamine plus amodiaquine (Spaq), is frequently used to prevent seasonal malaria in children during months of peak transmission periods such as rainy seasons. Previously, demand for drugs such as Spaq in Africa has been met through the importation of generic versions of the medicine from India and China.